A Bacterial CARD-NLR-like Immune System Controls the Release of Gene Transfer Agents
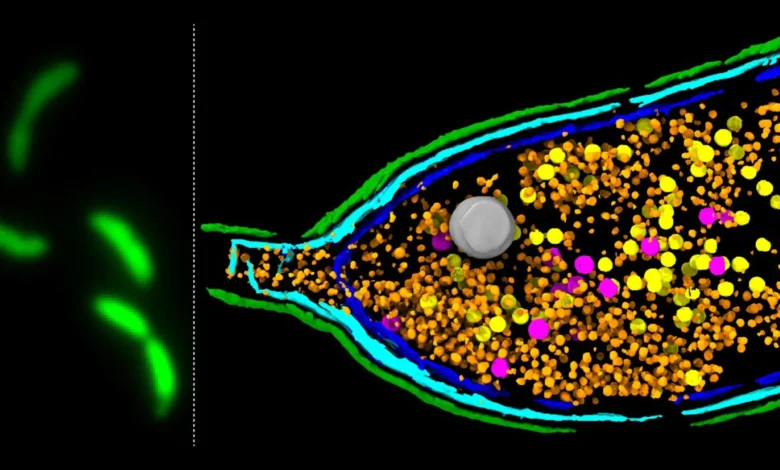
Scientists have unveiled groundbreaking insights into the intricate mechanisms by which bacteria share genes, including those that confer antimicrobial resistance (AMR), a burgeoning global health crisis. The pivotal findings, emerging from research conducted at the esteemed John Innes Centre, illuminate the sophisticated roles of unusual particles known as gene transfer agents (GTAs). These microscopic couriers, once thought to be mere viral relics, are now understood to be instrumental in the rapid dissemination of traits that threaten human health, particularly antibiotic resistance.
The Dual Nature of Gene Transfer Agents
At first glance, GTAs bear a striking resemblance to bacteriophages, the viruses that specifically infect bacterial cells. However, the research clarifies that GTAs are not hostile invaders. Instead, they are descendants of ancient viruses that bacteria have ingeniously adapted and brought under their own cellular command. This domestication process has transformed them from potential pathogens into sophisticated tools for bacterial evolution and adaptation.
These particles function as highly efficient delivery vehicles. They meticulously package fragments of DNA originating from their host bacterium and then transport these genetic payloads to neighboring bacterial cells. This fundamental biological process, termed horizontal gene transfer (HGT), is a cornerstone of bacterial diversity and adaptability. It allows bacterial populations to rapidly acquire and disseminate advantageous traits, such as the ability to withstand the onslaught of antibiotic medications, a critical factor in the escalating AMR crisis. The World Health Organization has repeatedly flagged AMR as one of the top 10 global public health threats facing humanity, estimating that by 2050, it could cause 10 million deaths per year.
Unlocking the Mystery of GTA Release
A critical, yet long-elusive, step in the GTA lifecycle is host cell lysis—the process by which the bacterial cell ruptures, releasing the newly formed GTA particles laden with genetic material. For years, the precise molecular machinery governing this escape remained a significant enigma for researchers. Understanding this mechanism is paramount, as it directly influences the efficiency and speed of gene transfer within bacterial communities.
The recent study, published in the prestigious journal Nature Microbiology, employed a cutting-edge deep sequencing-based screening methodology. This innovative approach allowed the researchers to meticulously identify and pinpoint the specific genes responsible for orchestrating GTA activity within the model bacterium Caulobacter crescentus. This bacterium, a common inhabitant of freshwater environments, serves as a valuable model for studying fundamental bacterial processes due to its well-understood genetics and rapid growth cycle.
The LypABC Control Hub: A Repurposed Immune System
The investigation yielded a significant discovery: a tightly regulated three-gene system, dubbed LypABC, which encodes bacterial proteins essential for GTA release. Experiments demonstrated a clear correlation between the presence and activity of these genes and the ability of the bacterial cell to lyse. When the lypABC genes were experimentally removed (deleted), the bacterial cells were rendered incapable of breaking open and releasing GTA particles. Conversely, when the LypABC system was deliberately overactivated, a substantial proportion of the bacterial population underwent lysis, confirming its role as the central control mechanism for GTA particle release.
Perhaps the most surprising and significant revelation of the study is the striking similarity between the LypABC system and a known bacterial defense mechanism: the anti-phage immune system. This bacterial immune system typically employs specific protein components to detect and neutralize invading bacteriophages. However, in the context of GTAs, this same molecular machinery appears to have been repurposed. Instead of defending against viral invaders, the bacteria are utilizing this evolved system to facilitate the release of their own gene-carrying particles, thereby promoting the spread of genetic information within their species. This remarkable example of biological repurposing underscores the immense evolutionary flexibility of bacteria.
This collaborative research, which involved significant contributions from the University of York and the Rowland Institute at Harvard, highlights a fundamental principle in biology: the reuse and adaptation of existing cellular systems for novel functions. It suggests that evolutionary innovation often builds upon pre-existing structures and pathways, rather than inventing entirely new ones.
The Imperative of Tight Regulation
The researchers also identified a crucial regulatory protein that plays a vital role in maintaining strict control over GTA activity. This oversight is not merely a matter of biological efficiency; it is a critical survival mechanism for the bacterial cell. Improper or excessive activation of the LypABC system can lead to uncontrolled cell lysis, which is highly toxic and detrimental to the bacterial population. This regulatory protein acts as a safeguard, ensuring that GTA release occurs only when and where it is beneficial for the bacterial community, preventing self-inflicted damage.
By unraveling the intricate workings of bacterial gene transfer, this study provides profound insights into the dynamic and flexible nature of bacterial biology. The ability of bacteria to share genes so readily and efficiently is a primary driver behind the alarming spread of antibiotic resistance. Understanding these mechanisms is therefore a crucial step in developing effective strategies to combat this global health threat.
Implications for the Fight Against Antimicrobial Resistance
Dr. Emma Banks, the first author of the study and a Royal Commission for the Exhibition of 1851 Research Fellow, emphasized the significance of the findings. "What’s particularly interesting is that LypABC looks like an immune system, yet bacteria are using it to release GTA particles," she stated. "It suggests that immune systems can be repurposed to help bacteria share DNA with each other—a process that can contribute to the spread of antibiotic resistance."
The implications of this research extend far beyond basic microbiology. For instance, the World Health Organization’s Global Action Plan on Antimicrobial Resistance (2015-2030) emphasizes the need for increased research and development into new antibiotics and alternative therapies. Understanding the fundamental processes of AMR gene spread, such as HGT mediated by GTAs, is crucial for designing interventions that can disrupt these pathways.
The next frontier for this research team is to delve deeper into the activation triggers of the LypABC system. Specifically, they aim to understand the precise molecular signals that initiate the process and how these signals ultimately orchestrate the rupture of bacterial cells and the subsequent release of GTA particles. This knowledge could pave the way for novel approaches to inhibit GTA-mediated gene transfer, thereby slowing the evolution and spread of antibiotic-resistant bacteria.
The journey of scientific discovery in this area has a history. Early observations of gene transfer in bacteria date back to the mid-20th century with the discovery of transformation, transduction, and conjugation. However, the role of specialized particles like GTAs in facilitating this exchange, particularly in environmental settings, has only come into sharper focus in recent decades. This latest research builds upon decades of foundational work, adding a critical piece to the complex puzzle of bacterial communication and adaptation.
The identification of LypABC as a repurposed immune system component highlights a broader biological principle that may be at play in other bacterial processes. It suggests that many seemingly disparate cellular functions might be interconnected through the adaptation of common molecular toolkits. This perspective could revolutionize how scientists approach the study of bacterial pathogenesis and evolution.
Furthermore, the economic and societal burden of AMR is immense. According to a report by the Pew Charitable Trusts, AMR could cost the global economy trillions of dollars annually if unchecked. The public health implications are equally dire, with the potential for common infections to become untreatable, leading to increased morbidity and mortality. Therefore, research that sheds light on the fundamental mechanisms of AMR spread, such as this study on GTAs, is not just academically significant but also critically important for global public health security.
The continued investigation into the LypABC system and its regulatory mechanisms promises to yield further insights. Understanding the specific environmental or cellular cues that trigger LypABC activation could reveal novel targets for antimicrobial drug development. For example, if specific nutrient limitations or stress signals are found to induce LypABC activity, strategies could be devised to mimic these conditions to promote GTA release, potentially making bacteria more susceptible to existing antibiotics or to treatments designed to disrupt HGT.
In conclusion, the research from the John Innes Centre has significantly advanced our understanding of how bacteria share genes, particularly those conferring resistance to antibiotics. By identifying the LypABC system as a key regulator of gene transfer agent release, and by highlighting its origins as a repurposed immune system, scientists have uncovered a crucial mechanism that contributes to the global AMR crisis. This work not only deepens our fundamental knowledge of bacterial biology but also offers new avenues for developing strategies to combat one of the most pressing health challenges of our time. The ongoing exploration into the intricacies of GTA activation and function is poised to provide even more critical clues in the ongoing fight against antimicrobial resistance.








