The Webb Lab: Unraveling the Mysteries of Adult Hippocampal Neurogenesis and Its Implications for Brain Health and Disease

For decades, a prevailing dogma in neuroscience asserted that the adult human brain possessed a fixed number of neurons, with no capacity for generating new ones. This long-held belief suggested that any neuronal loss, whether due to lifestyle choices, environmental toxins, or the natural aging process, was irreversible. However, this established doctrine has been significantly challenged by a growing body of research, particularly the work conducted in the laboratory of Associate Professor Ashley Webb at the Buck Institute for Research on Aging. Webb’s team is at the forefront of investigating neurogenesis, the process by which new neurons are formed, specifically within the adult hippocampus, a brain region critical for learning and memory.
Challenging the Dogma: New Frontiers in Human Neurogenesis
The notion that adult humans cannot produce new brain cells was largely based on observations in higher mammals. Yet, research in rodents demonstrably showed their ability to generate new neurons in specific brain areas through the activation of dormant neural stem cells. This discovery opened the door for scientists like Ashley Webb to explore whether a similar capacity might exist in the human brain.
"Although it remains somewhat controversial, tools have been developed over the past 10 years which make it possible to detect stem cells and new neurons in the human brain," Webb stated in a recent discussion about her lab’s focus. "These tools have really pushed the needle in terms of research."
This paradigm shift is not merely an academic curiosity; it holds profound implications for understanding and potentially treating age-related cognitive decline and neurodegenerative diseases like Alzheimer’s. The hippocampus, the primary site of adult neurogenesis in mammals, plays a pivotal role in forming and retrieving memories and in spatial navigation. Its degeneration is a hallmark of Alzheimer’s disease, leading to the characteristic memory loss and cognitive impairment associated with the condition.
The Hippocampus: A Hub for Learning, Memory, and Neurogenesis
Webb’s research highlights that current evidence points to the hippocampus as the sole region in the adult human brain where neurogenesis has been definitively observed. This concentration of new neuron formation in such a crucial area underscores its potential importance for maintaining cognitive function throughout life.
"Most of what we know about hippocampal neurogenesis is from mice and rats," Webb explained. "And in rodents, we know that neurogenesis is essential, particularly as the mice get older, in order to maintain optimal learning and memory related to little tasks that they do."
The critical questions now revolve around the functional significance of these newly generated neurons in humans. While the existence of neurogenesis in the adult human hippocampus is increasingly accepted, its precise role and contribution to cognitive health remain subjects of intense investigation.
Unraveling the Molecular Machinery of Neurogenesis
The Webb lab employs a highly mechanistic and molecular approach to dissect the intricate processes underlying neurogenesis. Their toolkit is extensive, encompassing genomics, transcriptomics, single-cell biology, metabolomics, proteomics, and advanced machine learning algorithms. By leveraging these cutting-edge technologies, the team aims to gain a comprehensive understanding of the molecular signals and pathways that govern the life cycle of a neuron, from its inception as a dormant stem cell to its integration into functional neural circuits.
"We’re focused on the rodent because we know it is a very well-characterized system that I think will inform what’s going on in humans," Webb elaborated. "And so, the work that we’re doing is really understanding the molecular mechanisms that support a neural stem cell and drive a neural stem cell from what’s essentially a dormant state in the hippocampus, all the way to a functionally integrated neuron."
This foundational research is crucial for developing targeted interventions. Understanding the precise molecular "on" and "off" switches for neurogenesis in healthy brains provides the essential blueprint for potentially reactivating or enhancing this process when it falters due to aging or disease.
Addressing Age-Related Decline in Neurogenesis
A central objective of the Webb lab is to understand how neurogenesis changes with age and to identify strategies for boosting it in older individuals. "We have two goals," Webb outlined. "One is to just understand the mechanisms that support healthy neurogenesis, and how those mechanisms change with age. And then we can start to focus on boosting things in the old mouse to actually get the stem cells to form neurons."
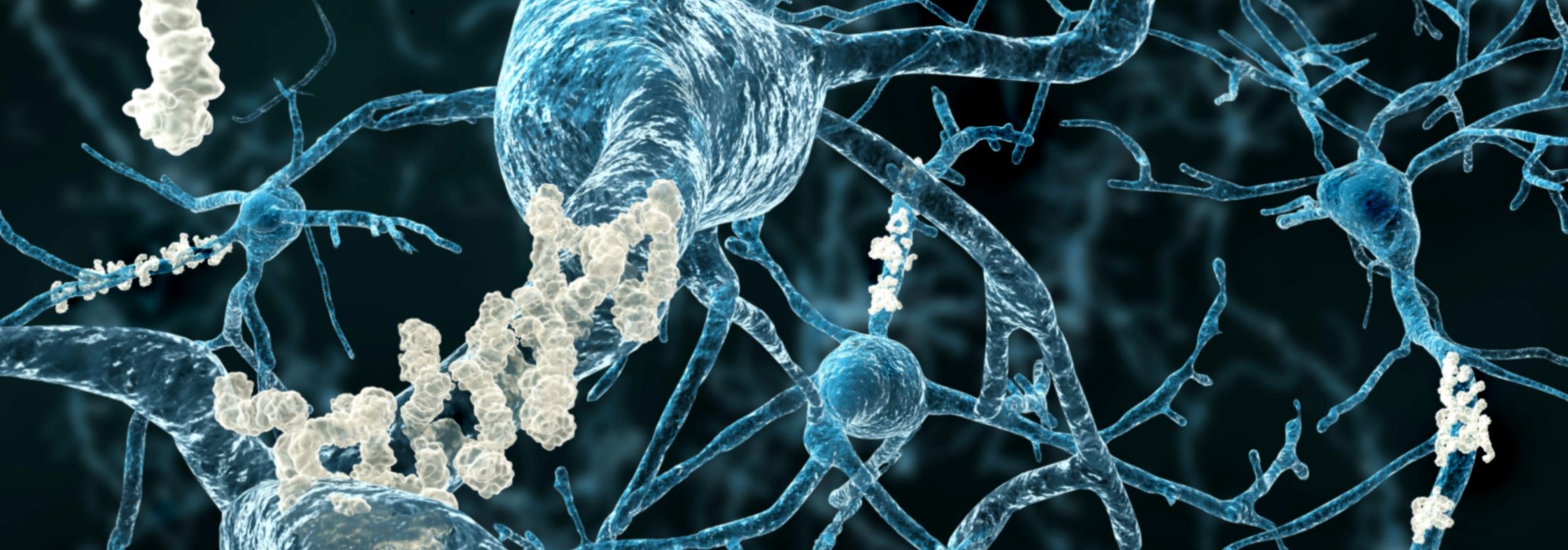
Progress has already been made in identifying age-associated changes that impair neurogenesis. The researchers have pinpointed deleterious alterations linked to faulty protein regulation and metabolic dysfunctions within aged neural stem cells. These findings suggest specific molecular targets that, when compromised, can reduce the generation of new neurons.
"We have some direct targets that we know decrease neurogenesis if you don’t have them," Webb stated. "Figuring out whether we can boost those targets is our next step."
Neurogenesis in the Context of Alzheimer’s Disease
The implications of the Webb lab’s work extend significantly to the study of Alzheimer’s disease. By investigating neurogenesis in mouse models of Alzheimer’s, the team seeks to determine if enhancing new neuron formation could offer a therapeutic avenue to improve hippocampal function in the face of neurodegeneration.
"We know neurogenesis declines in mouse models of Alzheimer’s disease," Webb confirmed. "And those tools I mentioned earlier now enable us to see the same phenomena in post-mortem brain samples from Alzheimer’s patients. So, decreased neurogenesis may be another problem that’s happening in the hippocampus and contributing to the cognitive decline."
While the extent to which reduced neurogenesis directly causes Alzheimer’s symptoms in humans is still under investigation, its observed decline in both animal models and human patients is a significant correlation. This suggests that restoring or preserving neurogenesis could be a complementary strategy to existing or emerging Alzheimer’s therapies.
The Rodent Model: A Crucial Foundation for Human Health
Webb emphasizes the indispensable role of rodent models in this research, despite the ultimate goal of understanding human brain function. The well-characterized nature of the rodent hippocampus, combined with the ability to manipulate genetic and environmental factors, makes it an ideal system for dissecting the fundamental biology of neurogenesis.
"I think it’s really important to do the basic research in mice, no matter what the levels of endogenous neurogenesis end up being in humans, because these dormant stem cells in the hippocampus are the only cells that we can tap into that normally make neurons in this context," she argued. "The scaffolding is there for the new neurons to arise from the stem cells, and the circuitry is there for the neurons to incorporate. And, importantly, the hippocampal stem cells are located in an area where there’s extensive neurodegeneration in Alzheimer’s disease. So, to me working in mice, and mouse models of disease, is the ideal context to understand how to make new neurons in any situation."
This perspective underscores a critical principle in biomedical research: a deep understanding of fundamental biological processes in well-established model organisms is often a prerequisite for translating discoveries to human therapies.
A Conceptual Framework for Future Therapies
The research conducted by the Webb lab contributes not only to specific findings but also to a broader conceptual framework that guides the entire field of neurogenesis research. Scientists worldwide are exploring various strategies to induce neurogenesis, including cellular reprogramming techniques and cell transplantation therapies.
"There are all kinds of people trying all different strategies to make new neurons, whether it’s cellular reprogramming methods, or cell transplantation where you deliver stem cells to different parts of the brain," Webb acknowledged. "I don’t know what’s going to work in the long run, but I think that understanding how a neuron forms in this context, this is our foundation. We have to understand the basic biology underlying neurogenesis in the mouse brain regardless of what’s going on in the human."
This foundational understanding of the molecular and cellular mechanisms that govern the birth, survival, and integration of new neurons in the adult brain is paramount. It provides the essential knowledge base upon which more complex and potentially transformative therapeutic interventions for age-related cognitive decline and neurodegenerative diseases can be built. The Webb lab’s meticulous investigation into the intricate dance of stem cells and developing neurons in the hippocampus is laying the groundwork for a future where the adult brain’s regenerative potential can be harnessed to promote lifelong cognitive health.