Navigating the Fog: Unraveling the Complex Interplay of Menopause, Brain Health, and Misdiagnosis in Midlife Women
The experience of cognitive decline, pervasive fatigue, and emotional dysregulation during midlife is often dismissed or misdiagnosed as depression, obscuring the profound impact of menopause on women’s health. For many women in their late 40s and early 50s, the struggle to recall common names, maintain focus, or manage everyday stress escalates beyond typical age-related memory lapses, manifesting as a debilitating "brain fog" that significantly impairs daily functioning and professional performance. This often leads to a protracted and frustrating search for answers within a healthcare system frequently ill-equipped to identify and treat the complex physiological shifts of the menopausal transition.
A Personal Journey Through Cognitive Impairment
The journey of one woman, grappling with increasingly severe cognitive and emotional symptoms, illustrates this critical gap in medical understanding. Her anecdotes paint a vivid picture: simple conversations, such as deciding on a restaurant, became arduous tasks. A typical exchange might involve a frustrating attempt to recall the name of a familiar eatery, leading to a convoluted description based on fleeting details—"that place with the peanut shells on the floor," "next to… you know… on that road where we used to take the dog to the vet." Hours later, the elusive name, "Texas Roadhouse!" would surface, a testament to the delayed retrieval plaguing her mind.
This "sludginess" with proper nouns, while sometimes associated with middle age, escalated into a far more debilitating condition for her. Beyond struggling with names of restaurants, people, books, or movies, her cognitive capacity waned significantly during workdays. Staring at a computer screen, she would find herself unable to initiate constructive work, her mind clouded by a haze akin to the semi-conscious state between sleep and full wakefulness. While mornings might offer a brief window of clarity, allowing her to condense hours of work into a mere two or three, her worst days began with an unshakeable mental fog, rendering work, reading, or any demanding activity impossible. This profound lack of mental bandwidth extended beyond professional tasks, impacting her ability to engage with daily life.
The Quest for Medical Clarity and Repeated Misdiagnosis
Alarmed by these persistent and worsening symptoms, she sought medical advice, initiating a two-year odyssey through various healthcare professionals. The initial recommendations were consistently antidepressants, a common response to symptoms like fatigue, low mood, and lack of motivation. However, trial after trial with different antidepressants, and even increased dosages, only exacerbated her condition, leaving her feeling more like a "zombie." A sleeping pill, offered to address her disrupted sleep, similarly deepened her drugged sensation.
Further investigations, including tests for thyroid function and anemia, yielded no abnormalities. Desperate, she experimented with a myriad of supplements, "mushroom coffee," and any product marketed for cognitive enhancement, all without relief. The turning point arrived almost incidentally during a routine annual gynecological exam. A mention of vaginal dryness, a symptom often overlooked or downplayed, prompted her gynecologist to inquire about a broader range of symptoms: sleep quality, mood, energy levels, hot flashes, and, crucially, brain fog.
Her response, delivered in a "hazy monotone," was poignant: "Funny you should mention brain fog… I feel like I’m barely alive." This pivotal conversation led to a critical realization: her symptoms were not indicative of clinical depression, but rather a manifestation of the menopausal transition. She was subsequently prescribed estradiol and progesterone, initiating a swift and dramatic turnaround. Within days, the mental fog lifted, her ability to think, type, follow conversations, and work past noon returned. Significantly, she experienced uninterrupted sleep for the first time in years, a profound relief after a prolonged battle with nocturnal awakenings. This personal account underscores the critical need for a more holistic and informed approach to women’s health during midlife.
Understanding Menopause: A Life Stage, Not a Disease
Menopause is not a disease or a medical condition but a natural and inevitable life stage, marking the end of a woman’s reproductive years. It is clinically defined as 12 consecutive months without a menstrual period. The preceding period, known as perimenopause, can last for several years, characterized by fluctuating hormone levels, primarily estrogen and progesterone, which trigger a wide array of symptoms. While common menopausal symptoms like weight gain and reduced libido often receive considerable attention, the cognitive and emotional impacts are equally significant, yet frequently overlooked or misattributed.
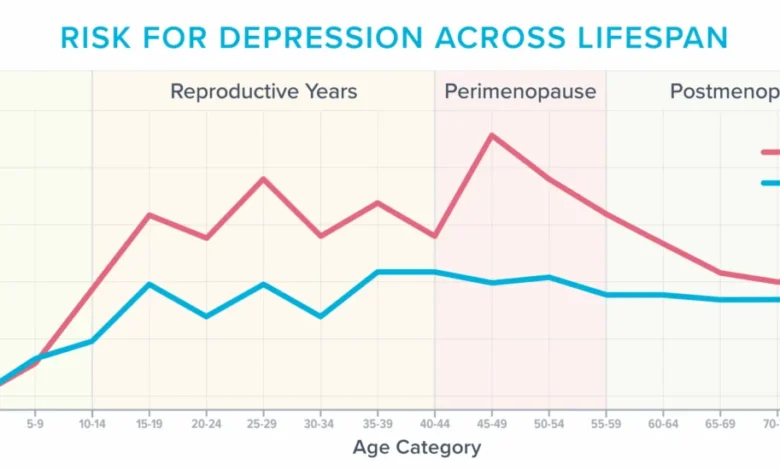
Research indicates that approximately 40% of women experience increased irritability, mood swings, anxiety, fatigue, and difficulty concentrating during and after menopause. This period also represents one of the most vulnerable times in a woman’s life for developing depression, especially for those with a history of mood disorders. The decline in estrogen, a neuroprotective hormone, is believed to play a significant role in these changes. Estrogen influences neurotransmitters, brain structure, and energy metabolism, and its reduction can lead to altered brain function.
The emotional landscape of menopause is complex. Many women report crying spells without apparent reason, heightened sensitivity to sensory stimuli—where normal sounds become physically painful—and increased anxiety in previously unbothersome situations, such as driving over bridges or through construction zones. These experiences highlight the systemic impact of hormonal changes on the central nervous system.
The Multifaceted Causes of Cognitive and Emotional Distress
The precise mechanisms driving these cognitive and emotional symptoms are not entirely clear but are understood to involve a combination of fluctuating hormone levels, typical age-related changes in brain structure, and significant life stressors often concurrent with midlife. Women in their 40s and 50s frequently find themselves at the peak of their careers, shouldering immense professional responsibilities that extend into their personal lives. Concurrently, many are navigating the challenges of parenting teenagers, caring for aging parents, adjusting to an empty nest, evaluating marital relationships, or managing significant financial burdens related to college education or healthcare costs. These cumulative stressors can profoundly deplete cognitive capacity and emotional reserves.
However, beyond these well-recognized factors, emerging research points to a less understood but critical trigger for cognitive and emotional distress: vasomotor symptoms (VMS), commonly known as hot flashes and night sweats.
The Anatomy and Impact of Vasomotor Symptoms
Vasomotor symptoms, characterized by sudden sensations of intense heat, sweating, and often palpitations, are more than mere inconveniences. They involve the constriction and dilation of blood vessels, impacting blood pressure and thermoregulation. During a hot flash or night sweat, there is a measurable rise in norepinephrine and cortisol levels, increased blood vessel dilation to dissipate heat, and an elevation in blood pressure and heart rate. Symptoms can range from skin reddening and warmth spreading across the face, neck, and chest, to sweating, heart palpitations, anxiety, fatigue, or faintness.
While the exact cause of hot flashes remains under investigation, one prominent theory suggests that declining estrogen levels disrupt the hypothalamus, the brain region responsible for temperature regulation. This disruption leads to a "wonky" internal thermostat, causing the brain to misinterpret the body’s temperature, triggering a physiological response to cool down, even when unnecessary.
For many years, hot flashes were largely considered benign, albeit embarrassing, symptoms. The personal account above reveals this perspective, as the author never thought to mention her hot flashes during numerous medical consultations. However, a growing body of research now highlights that VMS may have more profound and detrimental effects on both blood vessels and brain health, leading an increasing number of experts to classify them as a treatable medical condition.
Vasomotor Symptoms and Brain Changes
Recent studies have shed light on the neurological implications of frequent hot flashes. One significant study involving 226 women utilized monitors to track hot flash occurrences, alongside magnetic resonance imaging (MRI), sleep diaries, and smartwatches to record nocturnal awakenings. Researchers observed a higher prevalence of "whole-brain white matter intensities" – patchy areas in the brain – in women who experienced the most hot flashes. These lesions, once attributed solely to normal aging, are now considered predictive of future cognitive decline. Individuals with an abundance of these brain lesions face double the risk of a dementia diagnosis and triple the risk of future stroke.
The link between VMS and brain lesions is believed to stem, in part, from adverse changes in the cerebral blood vessels. A three-year study of 492 women found that those with frequent hot flashes exhibited unhealthy changes in their blood vessels, including a reduced ability to dilate in response to increased blood flow. Other research has correlated frequent hot flashes with increased risks of hypertension, cardiovascular disease, and endothelial dysfunction, all of which compromise cerebral blood flow and contribute to cognitive issues.
The Critical Link: Sleep Disruption and Brain Health
Beyond direct vascular effects, frequent hot flashes profoundly impact the brain by severely disturbing sleep. Critically, many women, like the author, may not consciously attribute their sleep disturbances to hot flashes, instead assuming they suffer from general insomnia or sleep apnea. This is because night sweats are not always overtly "sweaty." The surge of cortisol and norepinephrine that jolts a woman awake may dissipate the intense heat of the flash before she fully registers it, leading to repeated, seemingly unprovoked awakenings throughout the night.
These chronic awakenings interfere with the brain’s vital processes of memory consolidation, toxin metabolism, and the storage of daily information. Persistent sleep loss can also lead to reduced connectivity in the hippocampus, a brain region crucial for learning and memory. Furthermore, sleep deprivation heightens the reactivity of the amygdala, the brain’s emotional center, making individuals more susceptible to stress, anxiety, irritability, frustration, and even rage. These adverse brain changes can manifest within days to weeks of chronic sleep loss, underscoring the severe cumulative impact of years of disrupted sleep during menopause.

Barriers to Effective Care: The Systemic Challenges
The path to receiving appropriate care for menopausal symptoms is often fraught with systemic challenges. A significant issue lies in diagnostic overlaps. The Patient Health Questionnaire (PHQ-9) depression scale, a widely used tool, includes symptoms such as trouble sleeping, fatigue, changes in appetite, and difficulty concentrating – four items that directly mirror symptoms of menopause-related sleep deprivation. Checking these boxes can lead to a depression diagnosis, even when the underlying cause is the battle with sleep and hormonal shifts characteristic of menopause.
Compounding this problem is a pervasive lack of menopause-specific training within the medical community. Surveys reveal that 80% of medical residents feel "barely comfortable" discussing menopause, and few residency programs, including obstetrics and gynecology, offer comprehensive training in menopausal health. This educational deficit explains why many healthcare professionals fail to inquire about hot flashes or sleep disturbances when women present with fatigue, lack of drive, and inability to focus.
Moreover, even when vasomotor symptoms are clearly linked to cognitive and emotional distress, many healthcare professionals remain hesitant to prescribe Menopausal Hormone Therapy (MHT), often citing concerns about safety or risk. This reluctance, described by Michigan-based menopause-trained gynecologist Dr. Jerrold H. Weinberg as "defensive medicine," stems from a fear of litigation.
Re-evaluating Menopausal Hormone Therapy (MHT): Evidence-Based Insights
The widespread apprehension surrounding MHT largely originates from research conducted decades ago, particularly the Women’s Health Initiative (WHI) study, which linked certain types of hormones to a slightly increased risk of breast cancer and stroke. However, more contemporary research has refined this understanding, demonstrating that the perceived risks are highly dependent on several factors: the woman’s age, the specific dose and type of hormonal preparation, and the duration of hormone use.
Current expert consensus, including guidelines from the Menopause Society, indicates that for women under 60 and fewer than 10 years postmenopausal, the benefits of MHT often outweigh the risks, particularly for those experiencing moderate to severe menopausal symptoms. Furthermore, MHT offers significant health benefits beyond symptom relief, including a reduced risk of Alzheimer’s disease and osteoporosis, as confirmed by Dr. Weinberg. While antidepressants can alleviate some menopausal symptoms like mood swings and hot flashes, they come with their own side effect profiles and, from a "defensive medicine" perspective, may be seen as a "safer" alternative by some clinicians. However, they do not address the underlying hormonal deficiency or offer the systemic health benefits of MHT.
Empowering Patients: Advocating for Health
Given the complexities and challenges in obtaining appropriate care, women must become proactive advocates for their health. Dr. Weinberg and Helen Kollias, PhD, a physiology and molecular biology expert and science advisor at Precision Nutrition, offer critical advice:
- Seek Menopause-Trained Professionals: Prioritize healthcare providers who explicitly list menopause as a focus area in their bio or who hold certifications from organizations like the Menopause Society. The Menopause Society offers an online database to locate certified practitioners.
- Document Symptoms Meticulously: Keep a detailed log of symptoms, including their nature, frequency, severity, and any potential triggers. This documentation provides objective data, especially useful during appointments where brain fog or anxiety might impede clear communication. Track hot flashes (frequency, intensity, time of day), sleep patterns (duration, awakenings, quality), mood fluctuations, energy levels, and cognitive issues.
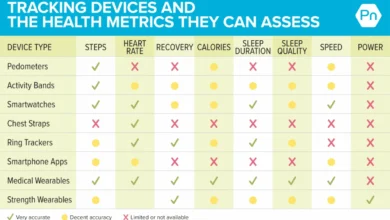
- Be Specific in Communication: Vague statements like "I don’t sleep well" are less effective than precise descriptions such as, "During the past seven days, I’ve had only one night of uninterrupted sleep lasting more than four hours. I wake, on average, five times per night, with my longest stretch of sleep being three hours." Utilize data from smartwatches or health apps to provide concrete evidence.
- Engage in Shared Decision-Making: Actively participate in discussions about treatment options, including the benefits and risks. Shared decision-making, a patient-centered approach, encourages clinicians to move beyond defensive medicine and collaborate with patients to make informed choices aligned with their values and circumstances. Ask direct questions: "What are the specific risks for me based on my health history and age?", "What are the potential benefits?", "Are there alternative treatments, and how do their risks and benefits compare?"
Holistic Lifestyle Strategies for Mental and Emotional Well-being
Beyond medical interventions, a comprehensive approach to managing menopausal cognitive and emotional symptoms includes fundamental lifestyle strategies that promote overall health. While there’s no magic bullet or special "menopause diet" (beyond avoiding known triggers like caffeine, alcohol, and spicy foods for some, and noting that soy products’ efficacy for VMS is less robust than once thought), foundational health habits become even more critical during this transition.
-
Lean into Fundamental Health Strategies:
- Nutrition: Prioritize a balanced, nutrient-dense diet rich in whole foods, vegetables, fruits, and lean proteins.
- Physical Activity: Engage in regular exercise tailored to your energy levels and recovery needs.
- Stress Management: Develop effective coping mechanisms for stress.
- Sleep Hygiene: Cultivate consistent and restorative sleep habits.
- Social Connectedness: Maintain strong social ties.
- Sense of Purpose: Engage in activities that provide meaning and fulfillment.
- Regularly assess these areas and identify barriers to consistent practice.
-
Experiment with Creatine: Beyond its known benefits for muscle and bone mass, creatine monohydrate (5-7 grams daily) may enhance mood, brain function, and reduce mental fatigue. It also shows promise in mitigating some negative effects of sleep deprivation.
-
Get Regular About Light Exposure: Exposure to natural light, particularly in the morning and late afternoon, helps regulate the circadian rhythm, promoting alertness during the day and better sleep at night. Studies show morning sunlight exposure correlates with improved sleep quality, faster sleep onset, longer sleep duration, and fewer awakenings. Sunlight also positively impacts mood and concentration.
-
Adjust Exercise Intensity: During midlife, recovery times increase, and injury risk rises. Intense, prolonged workouts can exacerbate fatigue and irritability, especially with poor sleep. Incorporate shorter, moderate sessions and prioritize recovery. Short bursts of movement (5-10 minute walks, quick sets of bodyweight exercises) can provide mental refreshes during the day. Gentle exercises like yoga or stretching can aid relaxation before bed, but avoid intense activity that releases adrenaline. Listen to your body, adjusting intensity and volume based on sleep and recovery, opting for Zone 2 cardio or reduced resistance training volume when needed.
-
Investigate Cognitive Behavioral Therapy for Insomnia (CBT-I): This evidence-based therapy teaches skills and cognitive reframing techniques to improve sleep. CBT-I helps establish consistent sleep schedules, regardless of prior night’s sleep quality, breaking cycles of poor sleep habits.
-
Get Real About Stress: Midlife demands often create an unsustainable burden. Critically evaluate responsibilities, tracking time and bandwidth usage. Identify areas for downsizing or delegating. Ask: "Is this essential?", "Can I automate or outsource this?", "Who can help me with this?" Tools like the "Wheel of Stress Assessment" can help pinpoint specific stressors. Learning to say "no" more often, without guilt, is crucial for preserving mental and emotional capacity.
-
Experiment with Cooling Technology: A cooler sleep environment can reduce night sweats and improve sleep quality. Try lowering the thermostat, using fans, or investing in cooling mattress pads.
-
Take Frequent Breaks: When brain fog sets in, productivity plummets. Permit short, restorative breaks (e.g., 20 minutes of quiet contemplation, listening to music, light stretching, or looking out a window). A 5-minute mind-body scan—focusing on physical sensations from head to toe without judgment—can provide a quick refresh and enhance self-awareness.
-
Follow a Diet Promoting Healthy Circulation: Diets that protect cardiovascular health also benefit brain health. The MIND and Mediterranean diets, rich in vegetables, fruits, whole grains, healthy fats (olives, nuts), beans, and fish, are associated with reduced risks of Alzheimer’s disease and depression. Nitrate-rich foods like beets and dark, leafy greens can temporarily improve memory by promoting blood vessel dilation and increasing blood flow to the brain.
The Hidden Opportunity of Menopause
While the challenges of menopause can be profoundly frustrating, this life stage presents a unique opportunity for introspection and re-evaluation. The diminishing capacity and relentless fatigue can force women to critically assess their priorities and ask fundamental questions about how they truly want to allocate their limited mental and physical resources.
For the woman whose story initiated this discussion, the period of cognitive impairment, before hormonal treatment, compelled her to confront an existential question: "Do I really need to be doing this?" This inquiry extended beyond her career, prompting a re-evaluation of daily commitments. She realized that working eight hours a day was not an absolute necessity, but rather a choice she could modify. Similarly, the expectation to cook elaborate dinners every night could be simplified or delegated. Crucially, learning to say "no" more often, and without regret, became a powerful tool for preserving energy for the things that truly mattered: spending time with aging parents, connecting with her college-aged child, and enjoying leisurely walks with friends.
Thanks to a combination of hormonal therapy and deliberate lifestyle adjustments, she regained her energy and mental clarity. Yet, she consciously chooses to end her workday around 3 p.m., not out of necessity, but by choice. This newfound ability to prioritize personal well-being and purposeful engagement over relentless productivity represents a significant, often unexpected, upside of navigating the menopausal transition. It underscores the importance of recognizing and treating menopausal symptoms, not just for individual well-being, but for empowering women to continue thriving and contributing meaningfully in all aspects of their lives.
References
Click here to view the information sources referenced in this article.