Global Readiness at a Crossroads: Six Years Post-COVID-19 PHEIC, WHO Warns of Fragile Progress in Pandemic Preparedness

Six years ago, the Director-General of the World Health Organization (WHO) invoked the highest global alert mechanism then available under international law, declaring the outbreak of a novel coronavirus disease – later identified as COVID-19 – a Public Health Emergency of International Concern (PHEIC). While the formal PHEIC status was rescinded in May 2023, the indelible impact of COVID-19 remains deeply etched in the collective global memory, continuing to resonate across societies and economies worldwide. As the world marks this six-year milestone since the initial declaration, the WHO has posed a critical question to countries and partners, a question it also directs inward: Is the world genuinely better prepared for the inevitable next pandemic? The answer, a complex blend of progress and persistent vulnerability, is both yes and no.
On one hand, there is a resounding "yes." Significant, concrete strides have indeed been made to bolster global preparedness frameworks. These efforts, catalyzed by the sheer scale and devastation of COVID-19, reflect an unprecedented level of international collaboration and investment in specific areas. However, this optimism is tempered by a stark "no." The progress achieved, while commendable, is characterized by its fragility and uneven distribution. Much more remains to be done, and sustained commitment is far from guaranteed, leaving humanity exposed to future health crises.
The Shadow of a Global Emergency: A Chronology of COVID-19’s Genesis and Evolution
The declaration of a Public Health Emergency of International Concern (PHEIC) on January 30, 2020, was a pivotal moment, signaling to the world the severe and potentially global threat posed by a previously unknown pathogen. A PHEIC, as defined by the International Health Regulations (IHR), is an extraordinary event which is determined to constitute a public health risk to other States through the international spread of disease and to potentially require a coordinated international response. This mechanism had been activated sparingly before COVID-19, notably for outbreaks such as H1N1 (2009), polio (2014), Ebola in West Africa (2014) and the Democratic Republic of Congo (2019), and Zika (2016). Each declaration served as an urgent call for heightened vigilance, resource mobilization, and coordinated action.
The initial emergence of the novel coronavirus, SARS-CoV-2, was first reported in Wuhan, China, in late 2019. By early January 2020, Chinese authorities had identified a new type of coronavirus, and the first confirmed cases began appearing outside China. The rapid person-to-person transmission and the severity of the illness prompted the WHO’s decisive action. The declaration of the PHEIC on January 30, 2020, was followed by a global escalation. On March 11, 2020, recognizing the uncontrolled international spread and significant impact, WHO Director-General Dr. Tedros Adhanom Ghebreyesus formally characterized COVID-19 as a pandemic.
The subsequent years witnessed an unprecedented global health crisis. The virus spread with terrifying speed, overwhelming healthcare systems, triggering lockdowns, disrupting supply chains, and causing immense economic dislocation. Globally, over 7 million deaths have been officially reported to the WHO, though the true mortality burden is widely believed to be significantly higher, potentially exceeding 20 million when accounting for excess deaths. The economic cost of the pandemic has been staggering, with estimates ranging into the tens of trillions of U.S. dollars in lost global GDP. Beyond mortality and economics, the pandemic left a legacy of long COVID, widespread mental health challenges, and exacerbated social inequalities. It fundamentally reshaped work, education, and social interaction.
After more than three years, during which global efforts led to the rapid development and deployment of vaccines, therapeutics, and diagnostic tools, the WHO declared an end to the PHEIC for COVID-19 on May 5, 2023. This decision reflected a significant decline in deaths, hospitalizations, and intensive care admissions, coupled with high levels of population immunity and improved disease management. However, the WHO was careful to emphasize that the virus itself had not disappeared and continued to pose a health threat, necessitating ongoing vigilance and integration of COVID-19 management into routine healthcare.
Tangible Strides in Global Health Security: Where Progress Has Been Made
The pandemic served as a brutal, yet invaluable, teacher. As Dr. Tedros highlighted at the 158th session of the Executive Board, "The pandemic taught all of us many lessons – especially that global threats demand a global response. Solidarity is the best immunity." This profound realization has underpinned a flurry of activities by the WHO, Member States, and various partners, leading to significant advancements in pandemic preparedness, prevention, and response.
One of the most critical areas of progress lies in the strengthening and proposed amendments to the International Health Regulations (IHR 2005). COVID-19 exposed significant gaps in the IHR’s implementation, particularly regarding timely information sharing, equity in access to countermeasures, and the enforcement of international obligations. Negotiations are currently underway to amend the IHR to address these shortcomings, aiming for more robust surveillance, faster notification mechanisms, and clearer responsibilities for Member States during health emergencies. Complementing this, an ambitious initiative to forge a new Pandemic Accord (or Treaty) is also in advanced stages of negotiation. This legally binding agreement aims to foster greater international cooperation, ensure equitable access to vaccines, diagnostics, and therapeutics, strengthen global governance, and establish a framework for pathogen access and benefit-sharing. Its adoption would mark a monumental shift towards a more just and effective global health security architecture.
Enhanced Surveillance and Early Warning Systems have also seen substantial improvements. The pandemic underscored the necessity of rapid pathogen identification and genomic sequencing. Investments have flowed into expanding laboratory capacities worldwide, improving real-time data sharing platforms, and establishing specialized intelligence hubs like the WHO Hub for Pandemic and Epidemic Intelligence in Berlin. These initiatives aim to detect new threats faster, track their evolution, and inform public health interventions with greater precision.
The acceleration of Research and Development (R&D) for vaccines and therapeutics was unprecedented during COVID-19. The rapid development of mRNA vaccines, for example, showcased the potential for scientific innovation under extreme pressure. Organizations like the Coalition for Epidemic Preparedness Innovations (CEPI) have gained renewed prominence, supporting the development of vaccines for known and unknown threats. Efforts are also underway to diversify manufacturing capabilities, particularly in low- and middle-income countries, to prevent future supply chain bottlenecks and ensure more equitable access to life-saving tools.
Furthermore, strengthening health systems at the national level has been a key focus. Many countries have invested in bolstering their primary healthcare infrastructure, training health workers, improving critical care capacities, and developing national emergency response plans. The lessons from COVID-19 have emphasized that a strong, resilient health system is the first line of defense against any health crisis.
The progress is not merely theoretical but demonstrably practical. Recent responses to outbreaks of highly virulent diseases like Ebola and Marburg serve as compelling evidence of improved national capabilities, supported by the WHO. A decade ago, the Ebola epidemic in West Africa (2014-2016) resulted in catastrophic loss of life, partly due to the lack of effective vaccines, rapid diagnostics, and limited treatment options. Since then, the landscape has been transformed. The most recent outbreaks of Ebola in the Democratic Republic of the Congo and Marburg in Rwanda, Tanzania, and Ethiopia were contained in a fraction of the time compared to previous epidemics, exhibiting limited spread and significantly lower case fatality rates. Crucially, these responses were led by national institutions, with WHO providing crucial technical, logistical, and operational support. This shift towards nationally-led, WHO-supported responses signifies a maturation of global health security efforts, reflecting stronger local capacities and faster, more coordinated international assistance.
These collective achievements are truly remarkable, embodying a shared global commitment to transcend national borders and sectoral divides. The overarching goal is to ensure that the world is never again caught unprepared by a pandemic and that no one is left behind in the response. Member States have made decisions that have not only enhanced the world’s capacity to respond rapidly and mitigate the impact of future pandemics but also, critically, to prevent them from emerging in the first place.
The Precipice of Progress: Why Gains Remain Fragile and Uneven
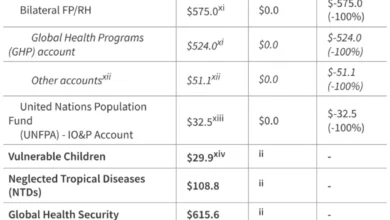
Despite these significant strides, the gains made in pandemic preparedness are perilously fragile. The past few years have been marked by profound turbulence in global health, and disconcertingly, a dangerous trend is emerging: funding priorities are shifting away from health and towards defense and national security. This reallocation of resources places at severe risk the very systems that were painstakingly strengthened during the height of the COVID-19 crisis to shield countries from future pandemics.
This reorientation of investment is profoundly shortsighted. Pandemics are not merely health crises; they are, unequivocally, national security threats. The notion that health security is distinct from national security is a perilous misconception. A robust national health system, capable of detecting, preventing, and responding to outbreaks, is as critical to a nation’s defense as its military. The economic devastation wrought by COVID-19, which led to an estimated $13.8 trillion in lost global output between 2020 and 2024 according to the International Monetary Fund, far surpasses the cost of proactive preparedness. Investing in preparedness is not a luxury; it is a fundamental investment in:
- Human Health and Well-being: Protecting citizens from illness, disability, and death.
- Economic Stability: Preventing widespread economic disruption, safeguarding livelihoods, and maintaining global trade.
- Social Cohesion: Preventing societal breakdown, maintaining public trust, and fostering community resilience.
- National Security: Protecting critical infrastructure, ensuring government functionality, and preventing geopolitical instability arising from health crises.
- Global Equity: Ensuring that all nations, particularly those with fewer resources, have the capacity to protect their populations.
The unevenness of progress further exacerbates this fragility. While some high-income countries have significantly enhanced their preparedness capabilities, many low- and middle-income countries continue to grapple with underfunded health systems, inadequate infrastructure, and chronic shortages of trained personnel. The stark inequities in vaccine access witnessed during COVID-19, where wealthy nations secured disproportionate supplies while others waited, serve as a potent reminder of how global health crises can deepen existing disparities and undermine solidarity. Unless preparedness efforts are truly universal and equitable, the world remains only as strong as its weakest link.
Furthermore, a phenomenon known as "pandemic fatigue" poses a significant threat. As the immediate crisis recedes, there is a natural human tendency towards complacency, a desire to revert to pre-pandemic priorities. This waning political will, coupled with competing global challenges such as climate change, geopolitical conflicts, and economic downturns, risks sidelining the long-term, sustained investment required for effective pandemic prevention. The memory of the acute crisis fades, but the pathogens do not.
The Indisputable Link: Health as National and Global Security
The argument that pandemics constitute national security threats is not abstract; it is empirically proven. Beyond the immediate health toll, a widespread disease outbreak can destabilize societies, disrupt governance, and even ignite international tensions.
Economically, pandemics halt production, cripple supply chains, and devastate industries like tourism and hospitality. The ensuing job losses and economic contraction can lead to social unrest and political instability. From a military perspective, outbreaks can incapacitate armed forces, disrupt deployment schedules, and compromise national defense capabilities. Moreover, the scramble for essential resources during a pandemic, such as personal protective equipment or vaccines, can strain international relations, leading to protectionist policies and a breakdown of trust.
The concept of "global health security" recognizes that no country can achieve health security in isolation. Pathogens respect no borders, and a threat emerging anywhere can rapidly spread everywhere. This interconnectedness necessitates a collaborative, multilateral approach, transcending traditional notions of national sovereignty. It demands sustained cooperation across sectors – health, finance, agriculture, environment, and defense – and across regions, to build resilient systems capable of anticipating, preventing, and responding to future threats.
Urgent Imperative: A Call to Action from the WHO and Beyond
Against this backdrop of fragile progress and persistent threats, the WHO has issued an unequivocal call to action to all governments, partners, and stakeholders: "Do not drop the ball on pandemic preparedness and prevention." This week’s WHO Executive Board meeting represents a pivotal moment in this ongoing journey. Member States are tasked with charting the course for the future of global health collaboration, accountability, and efficiency, defining roles and responsibilities in a complex and interconnected world.
The finalization and ratification of the Pandemic Accord and the amendments to the IHR are paramount. These instruments are designed to provide the legal and operational frameworks necessary for a more coordinated, equitable, and effective global response to future health emergencies. Their success hinges on the unwavering commitment and political will of all nations.
Beyond international agreements, the call to action encompasses several critical areas:
- Sustained and Predictable Funding: Governments must prioritize and allocate consistent, long-term funding for national and global health security initiatives, recognizing these as essential investments rather than discretionary expenditures.
- Strengthening National Health Systems: Focus must remain on building robust, resilient, and equitable health systems, particularly at the primary healthcare level, which serves as the frontline defense against outbreaks. This includes investing in infrastructure, equipment, and, crucially, a well-trained and supported health workforce.
- Investing in Research and Development: Continued support for innovative research into vaccines, diagnostics, and therapeutics for emerging pathogens is vital, alongside efforts to ensure equitable manufacturing and distribution capacities globally.
- Promoting Equity: All efforts must prioritize equity, ensuring that resources, tools, and expertise are accessible to all countries and populations, irrespective of their economic status. This includes addressing intellectual property barriers and promoting technology transfer.
- Fostering Multi-sectoral Collaboration: Recognizing the complex drivers of pandemics (e.g., climate change, deforestation, antimicrobial resistance), collaboration must extend beyond the health sector to include environment, agriculture, animal health (One Health approach), finance, and defense.
WHO remains steadfast in its commitment to working with all countries to strengthen preparedness, accelerate innovation, and uphold the principle of solidarity. The organization will continue to support Member States as they finalize their historic efforts to forge a global compact for a world that is fundamentally safer from pandemics.
The Path Forward: Sustained Vigilance and Collective Responsibility
The journey towards comprehensive pandemic preparedness is not a destination but an ongoing process. It demands continuous vigilance, adaptive strategies, and unwavering political commitment. The lessons from COVID-19 were hard-won, paid for in lives and livelihoods. To squander these lessons now, to allow complacency to creep in, would be a catastrophic failure of global leadership and collective responsibility. The time to prepare is not when the next pathogen emerges; the time to prepare is now, before the next pandemic strikes, ensuring that humanity is equipped with the tools, systems, and solidarity to face future threats with resilience and equity.