The HPV Vaccine: A Cornerstone of Cancer Prevention Amidst Evolving Policy and Public Health Challenges
The human papillomavirus (HPV) vaccine stands as a monumental achievement in public health, representing the first and only vaccination designed to prevent several cancers directly linked to various HPV strains. This groundbreaking medical innovation offers the profound promise of safely averting numerous forms of cancer attributable to HPV, diseases that have historically claimed the lives of countless individuals across all genders. Since its initial introduction to the U.S. market in 2006, the vaccine has undergone significant enhancements, now covering a broader spectrum of HPV strains, streamlining its dosage from three to two shots for most recipients, and crucially, achieving full cost coverage through both private insurance and public health programs. While initially recommended exclusively for girls and young women, its scope has since expanded to include boys, young men, and people of all genders, reflecting a comprehensive approach to population-level protection. Despite a general upward trend in vaccination rates over time, recent years have witnessed notable declines, particularly in the wake of the COVID-19 pandemic, signaling new challenges for public health initiatives. This comprehensive analysis delves into the intricate relationship between HPV and related cancers, the evolving landscape of HPV vaccine recommendations for all eligible populations, and the critical mechanisms ensuring insurance coverage and access.
Understanding HPV and Its Oncogenic Potential
Human papillomavirus is not merely a common infection; it represents the most prevalent sexually transmitted infection (STI) in the U.S., frequently acquired shortly after the initiation of sexual activity. The sheer scale of its presence is staggering, with approximately 42.5 million Americans currently infected and at least 13 million new infections occurring annually. Globally, the World Health Organization (WHO) estimates that nearly all sexually active men and women will acquire an HPV infection at some point in their lives, underscoring its ubiquitous nature. With more than 200 known strains, HPV presents a complex challenge. While the vast majority of HPV infections are transient and resolve spontaneously, persistent infection with high-risk strains can lead to cellular changes that, over time, may progress to cancer.
The burden of HPV-related cancers has surged dramatically over the past decade. Between 2018 and 2022, over 49,000 individuals in the United States were diagnosed with an HPV-related cancer, a significant increase from approximately 30,000 cases recorded in 1999. This rise is particularly concerning given shifts in the types of cancers observed. While HPV-related cervical and vaginal cancer rates have seen a welcome decrease since 1999, rates for oropharyngeal (throat) and anal HPV-related cancers have alarmingly increased, highlighting an evolving epidemiological landscape.
Cervical Cancer: Progress and Persistent Disparities

Cervical cancer remains one of the most significant HPV-related diseases, with over 90% of cases directly attributable to the virus. Specifically, two high-risk strains, HPV 16 and 18, are responsible for approximately 66% of cervical cancer cases worldwide. In the U.S., despite advancements in screening and treatment, an estimated 13,360 new cervical cancer cases were diagnosed in 2025, leading to approximately 4,320 deaths. Globally, cervical cancer continues to be a leading cause of cancer-related mortality among women, especially in low- and middle-income countries where access to screening and vaccination is limited.
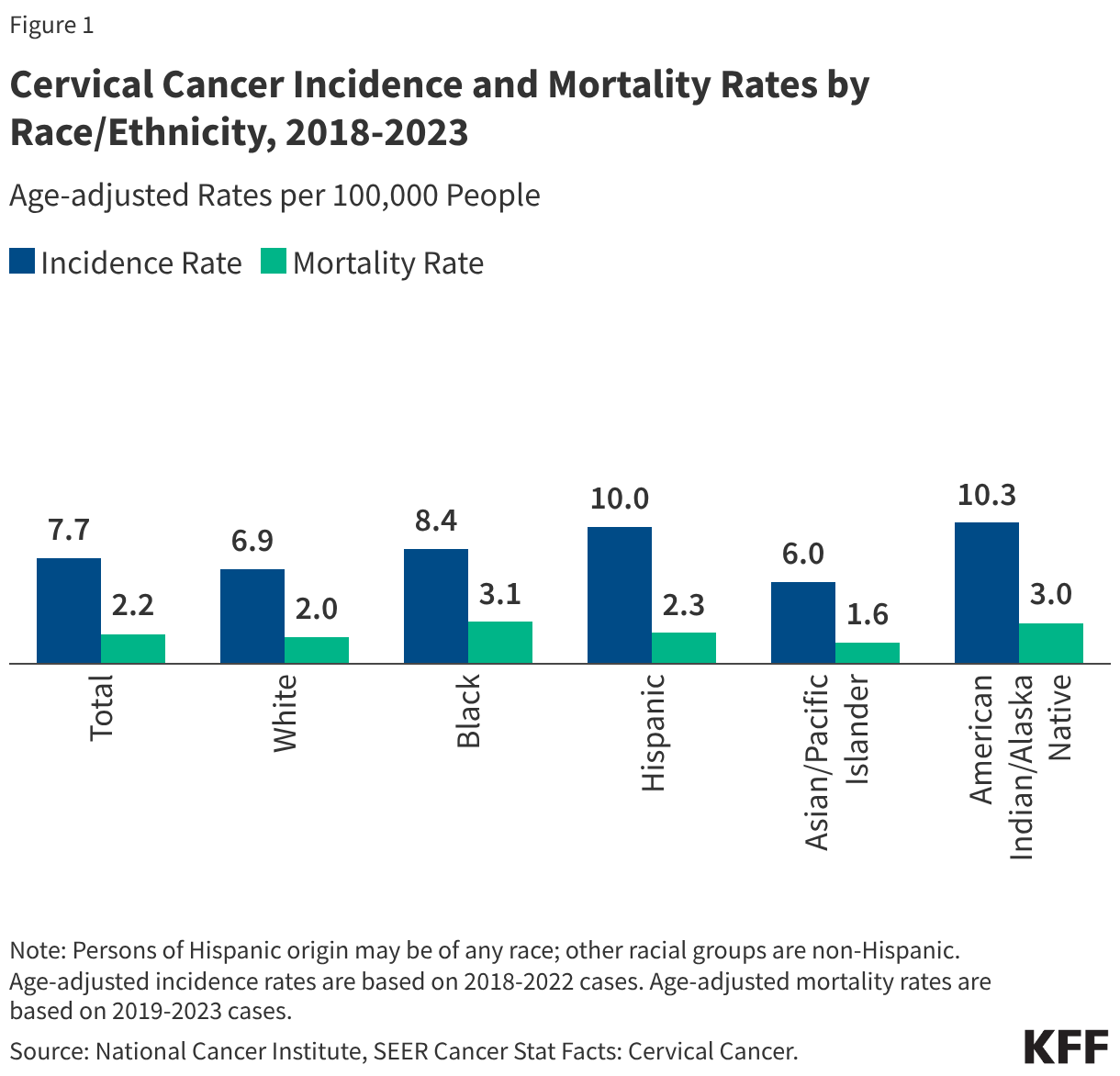
Despite the widespread availability of cervical cancer screening methods, racial disparities in both incidence and mortality rates persist within the U.S., revealing systemic inequities in healthcare access and outcomes. For instance, Hispanic women, who experience the second-highest incidence rate of cervical cancer, demonstrate mortality rates comparable to the national average. In stark contrast, Black women, while having the third-highest incidence rate, tragically suffer the highest mortality rates from the disease. This paradox is further underscored by data indicating that both Black and Hispanic women exhibit the highest rates of recent Pap testing, suggesting that screening alone is insufficient to close the mortality gap. Factors contributing to this disproportionate impact are multifaceted and deeply rooted in systemic issues, including lower rates of follow-up treatment after an abnormal screening result, disparities in access to optimal treatment options, diagnoses at later stages of disease progression, and pervasive negative experiences within the medical system that erode trust and hinder consistent care. These factors collectively illustrate the complex interplay of social determinants of health and structural racism in shaping health outcomes. The data represented in Figure 1 vividly illustrates these persistent disparities in cervical cancer incidence and mortality across different racial and ethnic groups from 2018-2023.
The Rising Tide of Oropharyngeal and Anal Cancers
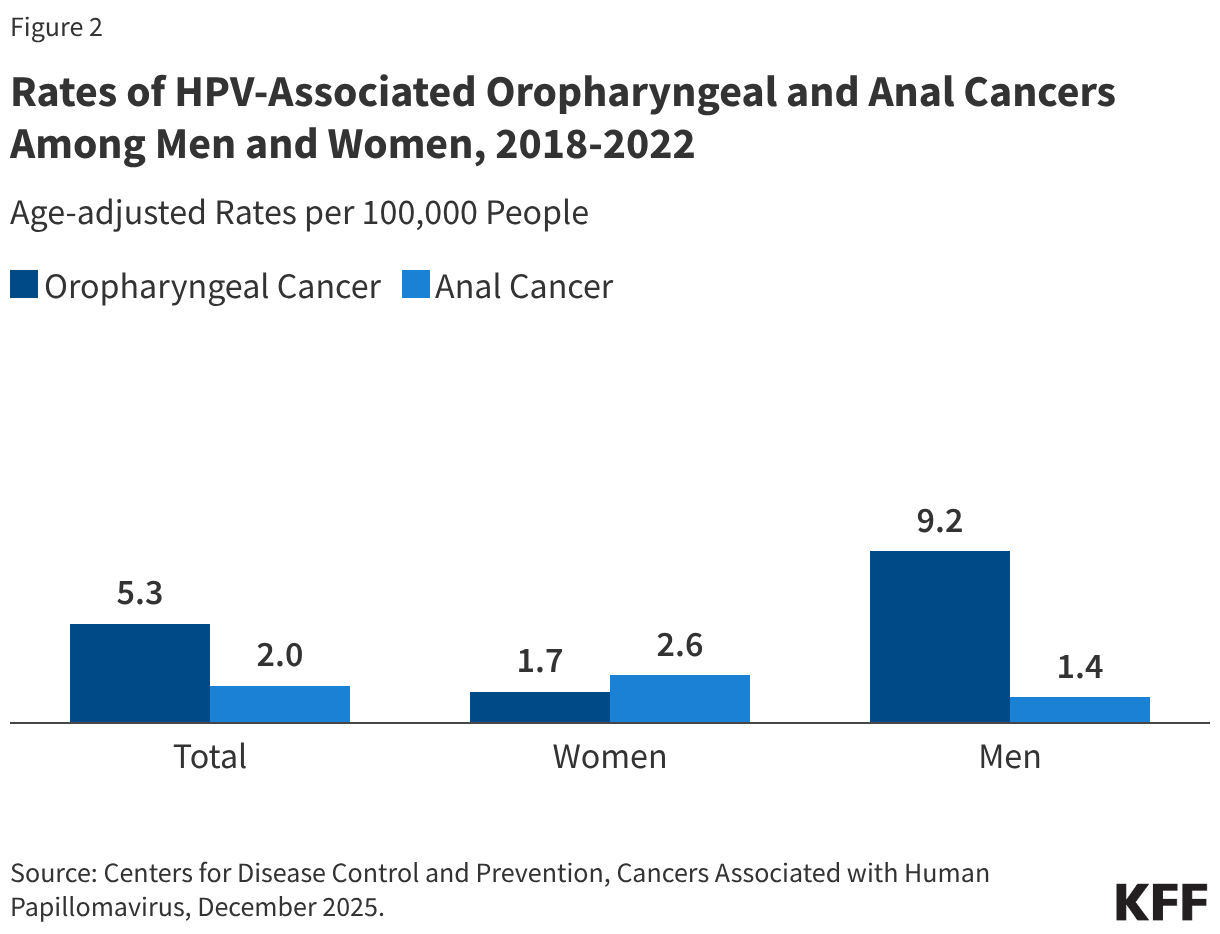
Beyond cervical cancer, HPV is a major causative agent for other increasingly prevalent malignancies. Approximately 22,585 cases of oropharyngeal (throat) cancer occur annually in the U.S., with an estimated 70% likely caused by HPV. These cancers are now the most common HPV-associated cancer among men and are generally more prevalent in men than women, as indicated in Figure 2. However, individuals who heavily use both tobacco and alcohol face a significantly elevated risk of developing these cancers, as these substances act synergistically with HPV. Research robustly suggests that HPV vaccines can effectively protect against these throat cancers, given their strong association with HPV 16 and 18, strains explicitly targeted by the vaccine.
HPV is also responsible for the vast majority (91%) of the estimated 7,600 annual cases of anal cancer in the U.S. While anal cancer cases are generally higher among women, men who have sex with men (MSM) face a disproportionately higher risk of developing anal cancer linked to HPV 16 and 18. Additional risk factors for anal cancer include a history of cervical cancer and a suppressed immune system. mirroring the trend in oropharyngeal cancers, there has been a notable increase in the rate of anal cancers over the past 15 years. Figure 2 provides a comparative view of the rates of HPV-associated oropharyngeal and anal cancers among men and women from 2018-2022, highlighting the gender-specific prevalence.
The Evolution of HPV Vaccination: From Development to Current Recommendations
The journey of the HPV vaccine began with rigorous scientific research into the virus’s role in cancer development, culminating in a groundbreaking medical innovation. Since 2016, Gardasil®9, manufactured by Merck, has been the sole HPV vaccine available in the U.S., representing the pinnacle of vaccine technology in this domain. The first-generation Gardasil® was approved by the FDA in 2006, offering protection against four HPV strains: 6, 11, 16, and 18. A significant leap forward occurred in December 2014 with the approval of Gardasil®9 for individuals aged nine to 45 years. This advanced vaccine protects against nine HPV strains (6, 11, 16, 18, 31, 33, 45, 52, and 58) that are associated with the majority of cervical, anal, and throat cancers, as well as most cases of genital warts and other HPV-associated anogenital diseases.
Initially, the vaccine’s approval focused on cervical cancer prevention. However, in 2020, the FDA broadened its approval to include the prevention of oropharyngeal cancer and other head and neck cancers, acknowledging the vaccine’s wider protective capabilities. Current global research emphatically demonstrates that Gardasil®9 protection is long-lasting, with more than 10 years of follow-up data in both boys and girls indicating sustained effectiveness and no evidence of waning protection. While the need for future booster shots remains an open question, the existing data is highly encouraging. International examples further bolster confidence: in Scotland, recipients of the bivalent HPV vaccine, Cervarix® (which protects against HPV 16 and 18), who were fully vaccinated at ages 12 or 13, have experienced no cases of cervical cancer since the vaccine program commenced in 2008. Moreover, recent data from the American Society of Clinical Oncology reveal that the vaccine has reduced the risk of all HPV-associated cancers, including oropharyngeal, head, and neck cancers, by a remarkable 50% in men.
Immunization recommendations for the U.S. population are typically issued by the federal Advisory Committee on Immunization Practices (ACIP), an expert body convened by the CDC. Historically, ACIP has comprised clinicians, scientists, public health experts, and other professionals with deep expertise in vaccine-related policies. In June 2024, during the Biden administration, ACIP recommended a two-dose series of the HPV vaccine for most adolescents. This recommendation strategically aimed to promote immunization during the period of maximal effectiveness – before the initiation of sexual activity. It is important to note that individuals already infected with HPV can still benefit from the vaccine, as it can prevent infection against HPV strains they may not have yet contracted, though it does not treat existing infections.
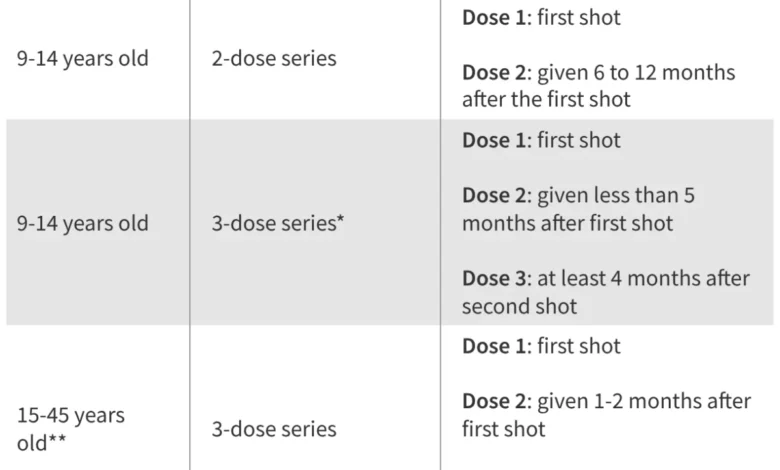
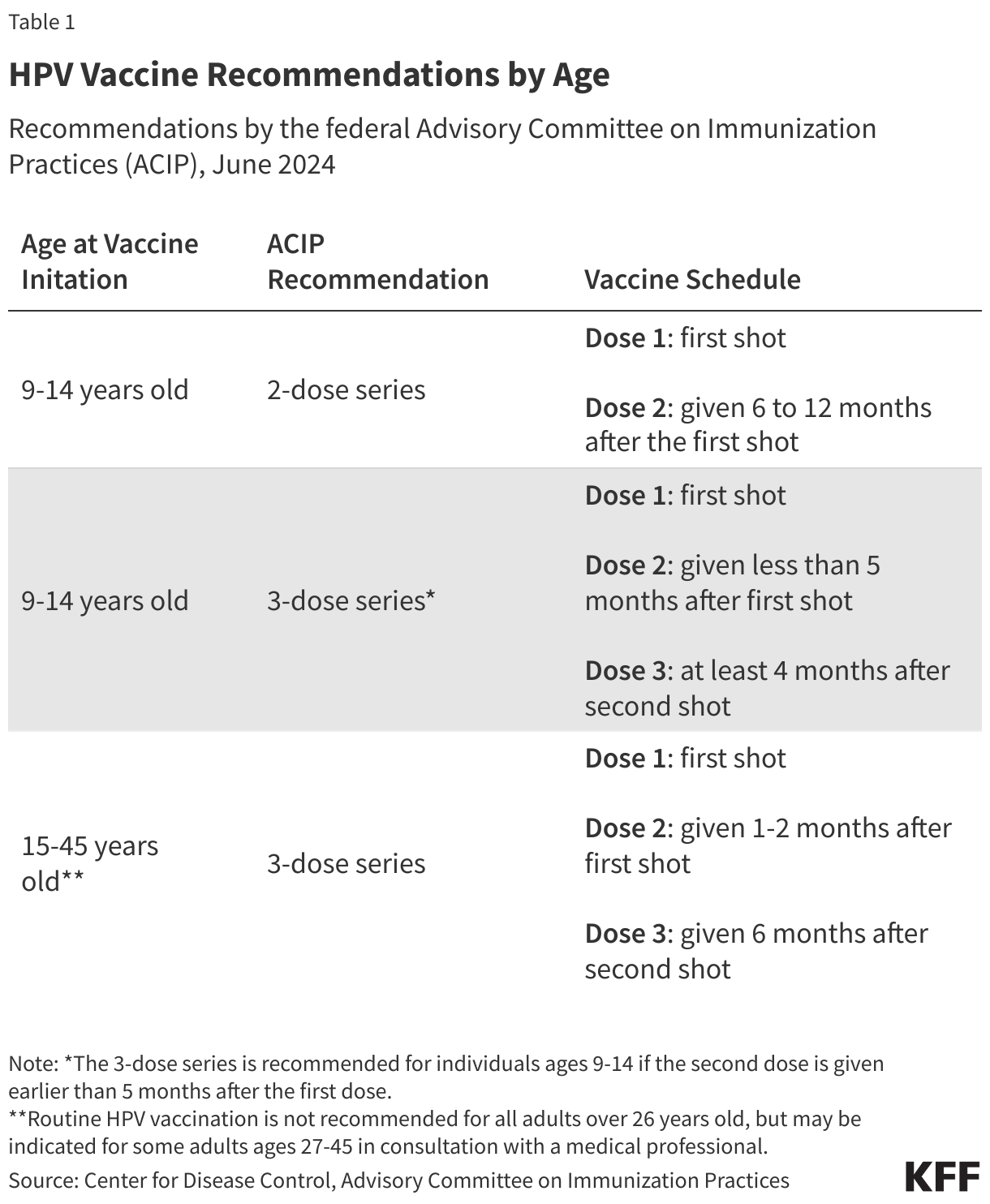
However, the trajectory of HPV vaccine recommendations faced an unprecedented disruption following the second Trump Administration taking office. In a move that sent shockwaves through the public health community, the Department of Health and Human Services (HHS) enacted broad changes in vaccine policy. In June 2025, Secretary of HHS, Robert F. Kennedy Jr., controversially dismissed the entire membership of ACIP, replacing them with new advisors, many of whom were known to hold skeptical views on vaccines. This reconstituted ACIP, in December 2025, subsequently altered the recommendation from a two-dose series to a single dose for adolescents. These revisions, alongside other pediatric vaccine recommendation changes made by the newly appointed committee, were swiftly challenged in a federal court by a coalition of public health and health professional organizations, prominently led by the American Academy of Pediatrics. The court’s ruling temporarily blocked these changes, including those to ACIP’s membership. Consequently, the 2024 recommendations, advocating a two-dose series for adolescents, currently remain in effect, pending further legal developments. Table 1 outlines the standard HPV vaccine recommendations by age, reflecting the current guidelines.
While the FDA has expanded its approval of the HPV vaccine to include adults aged 27 to 45, ACIP has not issued a blanket recommendation for routine catch-up vaccinations for all adults in this age group. Instead, ACIP advises that adults aged 27 to 45 who have not been adequately vaccinated and who may be at risk for new HPV infections should engage in a shared decision-making process with a medical professional to determine the appropriateness of receiving the vaccine. This nuanced approach acknowledges individual risk factors and the potential benefits for those not previously vaccinated.
HPV Vaccine Uptake: Progress, Gaps, and Barriers
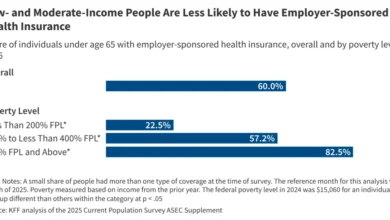
In 2024, more than 60% of adolescents aged 13-17 in the U.S. were considered up-to-date (UTD) with their HPV vaccinations, reflecting significant progress since the vaccine’s introduction. Analysis of demographic trends reveals interesting disparities: adolescents of Asian or Black descent, or those covered by Medicaid, were more likely to be HPV UTD compared to their White, privately insured, or uninsured counterparts. While HPV vaccination rates among teen boys have historically lagged behind girls, they have been steadily rising since 2016, reaching 61% HPV UTD for boys versus 64% for girls in 2024, narrowing the gender gap.
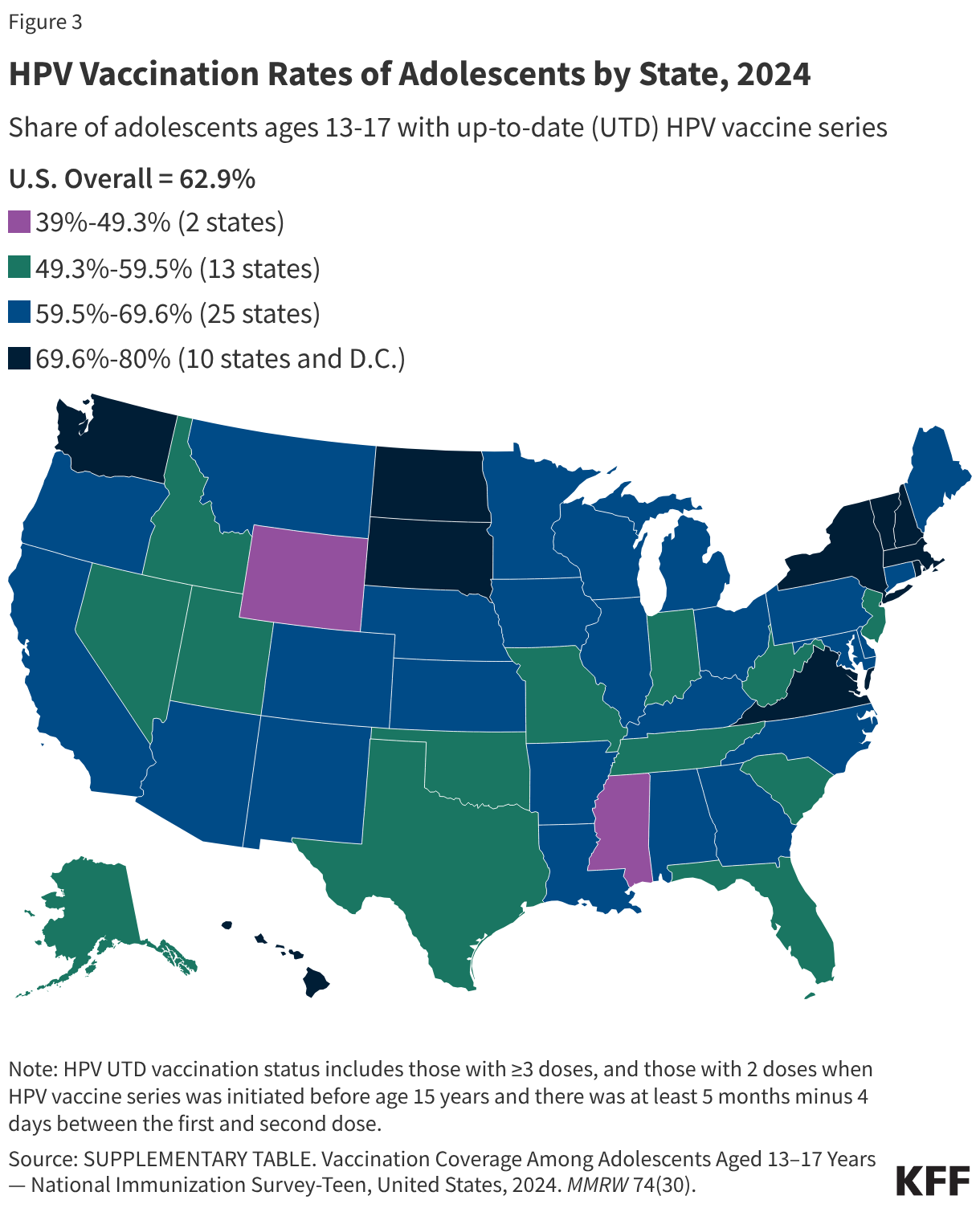
However, the geographic distribution of HPV vaccination rates across the U.S. remains highly variable. As depicted in Figure 3, state-level uptake ranges dramatically, from a low of 39% of adolescents being HPV UTD in Mississippi to a high of 80% in Massachusetts. This variability is often influenced by state-specific policies. Some states, including Hawaii, Rhode Island, Virginia, and the District of Columbia, have enacted laws that require HPV vaccination for school entry, demonstrating a proactive approach to public health. In contrast, states like California, through its Cancer Prevention Act, mandate schools to notify families of 6th-grade children about HPV vaccine recommendations but do not require adherence for school entry. Furthermore, most states permit vaccine exemptions based on religious or personal beliefs, which can contribute to lower coverage rates in certain areas.
A critical challenge in HPV vaccination is not just initiation but also completion of the full series. In 2024, while 79% of adolescent girls and 77% of boys received at least one dose of the HPV vaccine, a notable gap exists between initiation and being fully up-to-date. Recent trends indicate that overall HPV vaccination initiation has stalled for the third consecutive year, suggesting a plateau in efforts. Throughout the last decade, vaccination rates have consistently remained lower among adolescents residing in predominantly rural areas compared to those in urban settings, highlighting disparities in access or outreach. Data consistently show that children are significantly more likely to be vaccinated when their parent or guardian receives a direct vaccine recommendation from a healthcare provider. Furthermore, while vaccine initiation among adolescents overall remained steady, initiation rates in recent years have slightly declined among adolescents who are uninsured or covered by Medicaid, as illustrated in Figure 4. This decline among vulnerable populations is particularly concerning.
Vaccine hesitancy is a significant contributing factor to lower HPV vaccination coverage among these subgroups. Prior to the COVID-19 pandemic, parents’ most commonly cited reasons for not vaccinating their children included concerns about vaccine safety and the belief that the vaccine was not necessary. Since the onset of the pandemic, many healthcare providers have observed a marked increase in vaccine hesitancy or outright refusal among parents of adolescents. This shift is often attributed to difficulties and disruptions caused by COVID-19, as well as a broader erosion of trust in vaccines and public health institutions. The public skepticism of HHS Secretary Kennedy, coupled with the vacillating recommendations for the HPV vaccine during the second Trump administration, are likely to further exacerbate hesitancy and sow confusion among both parents and clinicians, posing a substantial threat to the progress made in cancer prevention.
Ensuring Access: Financing and Coverage Mechanisms
The availability of multiple public and private financing sources is crucial in ensuring that nearly all children and young adults in the U.S. have comprehensive coverage for the HPV vaccine. Many of these financing entities base their coverage policies on the evidence-based recommendations issued by ACIP.
The Affordable Care Act (ACA) has played a pivotal role in expanding access, requiring both public and private insurance plans to cover a wide array of recommended preventive services, including ACIP-recommended immunizations, without consumer cost-sharing. This mandate ensures that plans must cover the full cost of the HPV vaccine, as well as essential screening services like Pap tests and HPV testing for women, removing financial barriers to critical preventive care.
Public Financing Programs
- Vaccines for Children (VFC) Program: This federal program, managed by the CDC, purchases vaccines at a discounted rate and distributes them to participating healthcare providers nationwide. Children are eligible through age 18 if they are uninsured, underinsured, Medicaid-eligible, Medicaid-enrolled, or American Indian or Alaska Native, ensuring that financial status does not preclude vaccination for vulnerable populations.
- Medicaid: As a joint federal and state program, Medicaid covers ACIP-recommended vaccines for enrolled individuals under age 21 through its Early and Periodic Screening, Diagnostic, and Treatment (EPSDT) program. For adults aged 21 and older, Medicaid covers approved adult ACIP-recommended vaccinations without cost-sharing, extending protection across the lifespan for eligible beneficiaries.
- Public Health Service Act (Section 317): This vital provision of the Public Health Service Act provides grants to state and local agencies, empowering them to expand the availability of vaccines to uninsured adults across the United States. These funds are often strategically directed towards meeting the needs of priority populations, including underinsured children and uninsured adults, thereby addressing critical gaps in access.
- Children’s Health Insurance Program (CHIP): CHIP serves children in families whose incomes are too high to qualify for Medicaid but too low to afford private insurance. Each state administers its own CHIP program with specific qualification criteria, jointly funded by state and federal governments. CHIP programs that operate separately from Medicaid Expansion are mandated to cover ACIP-recommended vaccines for beneficiaries, as these children are not eligible for coverage under the federal VFC program.
Manufacturer Assistance
- Merck Vaccine Patient Assistance Program: Merck, the manufacturer of Gardasil®9, has established a dedicated assistance program to provide free HPV vaccines in the United States. To qualify for this program, individuals must be aged 19 or older, uninsured, and meet specific low-income criteria, offering a crucial safety net for adults who might otherwise face prohibitive costs.
Broader Implications and The Future of HPV Prevention
The HPV vaccine represents an unprecedented opportunity to eliminate several cancers, a goal that few medical interventions can realistically aspire to. Its development and widespread adoption could dramatically reshape the global cancer burden. However, realizing this full potential hinges on addressing significant challenges, particularly concerning policy instability and public trust. The political interference in ACIP’s independent, evidence-based process, and the subsequent legal battles, underscore a dangerous precedent that can erode confidence in public health recommendations. Such vacillation not only confuses the public but also places clinicians in a difficult position, potentially hindering their ability to effectively recommend and administer life-saving vaccines.
Sustaining and expanding HPV vaccine uptake requires a multi-pronged strategy. This includes vigorous public health campaigns to counter misinformation and educate communities about the vaccine’s safety and efficacy. Crucially, healthcare providers must be empowered and supported to consistently recommend the vaccine, serving as trusted messengers. Addressing access barriers, especially in underserved rural communities and among uninsured or Medicaid-covered populations, is paramount. This may involve enhancing mobile clinics, expanding school-based vaccination programs, and ensuring consistent funding for public health initiatives. Globally, vast disparities in vaccine access persist, particularly in low-income countries where cervical cancer mortality remains highest. International efforts must continue to support equitable distribution and implementation of HPV vaccination programs worldwide.
In conclusion, the HPV vaccine stands as a testament to scientific progress and a beacon of hope in the fight against cancer. Its journey, marked by scientific breakthroughs, expanding recommendations, and robust coverage mechanisms, has undeniably saved lives. Yet, recent challenges related to declining uptake and political interference threaten to undermine this progress. The imperative now is to reaffirm the commitment to evidence-based public health, protect the integrity of expert advisory bodies, and intensify efforts to ensure that every eligible individual has access to this transformative cancer-preventing vaccine, securing a healthier future for all.